
Clinical trials
Consolidation of Clinical trials
IRBLleida’s Clinical Research Unit is an initiative to consolidate trials with no commercial interest, enhance private clinical studies and strive for excellence and leadership in both.
Objectives
The unit aims to provide support for clinical research groups at the Arnau de Vilanova University Hospital and Santa Maria University Hospital for conducting clinical trials, especially those that have no commercial interest.
Scope
The unit's scope encompasses clinical drug trials, medical devices and other therapeutic interventions studies. The project also includes randomized clinical trials of intervention for diagnostic, preventive or service purposes.
Advice and support
The unit offers advice and support on issues related to methods and statistical, regulatory and procedural aspects, economic aspects and medication management, implementation tests, data management and results analysis. The advice and support includes very specialized aspects which are the responsibility of the researcher body such as those related to documenting, archiving, monitoring and follow-up clinical trials.
Contact
Clinical research groups can contact the unit by email afernandez(ELIMINAR)@irblleida.cat.
Clinical Research Support Unit (USIC)
Activities and Services
The ethical, methodological and legal evaluation of clinical trials and research projects must be carried out by the Medicines Research Ethics Committee (CEI/CEIm) of the Hospital Universitari Arnau de Vilanova de la Gència Territorial de Lleida i GSS. Spanish and European law on clinical trials requires a major development of faculties and functions of the Clinical Research Ethics Committee, above all with regard to multicentre trials.
Furthermore, the implementing requires that post-authorization observational studies be assessed by the Commiteee. The main objective is to contribute to improving the quality, management and relevance of clinical trials that aim to develop our environment. This is a clear commitment to strengthen the position of the Hospital and its Clinical Research Ethics.
Map of processes
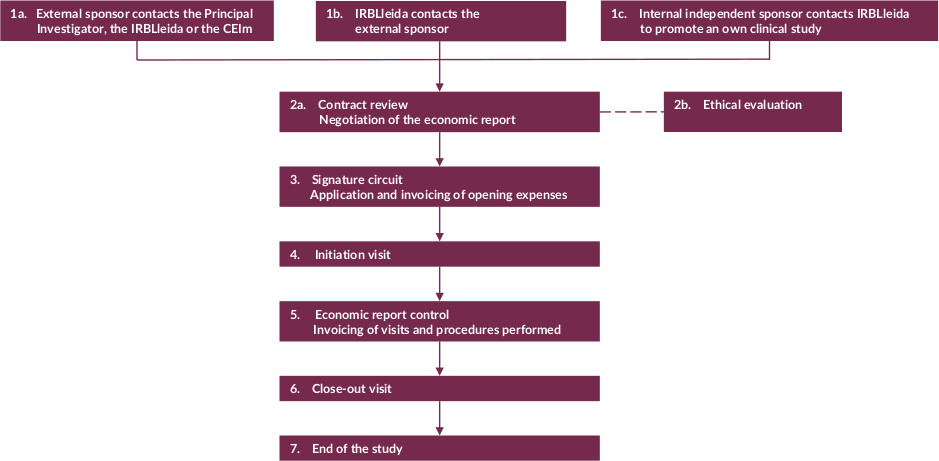
More informatiom:
- Rates (approved by the IRBLleida board of trustees on 13/06/2024)
- Fees CEIm HUAV (CAT)
- Overheads Policy






